[EOFLOW] EOFLOW, Giving Hope to Diabetes Patients
- Name 관리자
- Date 2021-12-24
▲Promotional Video
EOFLOW, Giving Hope to Diabetes Patients
Can you briefly introduce us to EOFLOW?
「Commercializing the world’s second, and Korea’s first disposable wearable insulin pump」
Since its founding in 2011, EOFLOW has become a company specializing in medical devices that leads the wearable drug delivery market. We have commercialized the world’s second, and Korea’s first tubeless, wearable and disposable “EOPatch” insulin pump. In April 2021, our “EOPatch” was officially introduced to the Korean market with Huons, and will soon be sold in the European market with European pharmaceutical company, Menarini. EOFLOW has subsidiaries in the United States and in Korea, and there are approximately a total of 120 employees worldwide including the headquarters. The company was listed on the KOSDAQ stock market in September 2020.
What are some features of the “EOPatch?”
「Improving user convenience, efficiency, and quality of life」
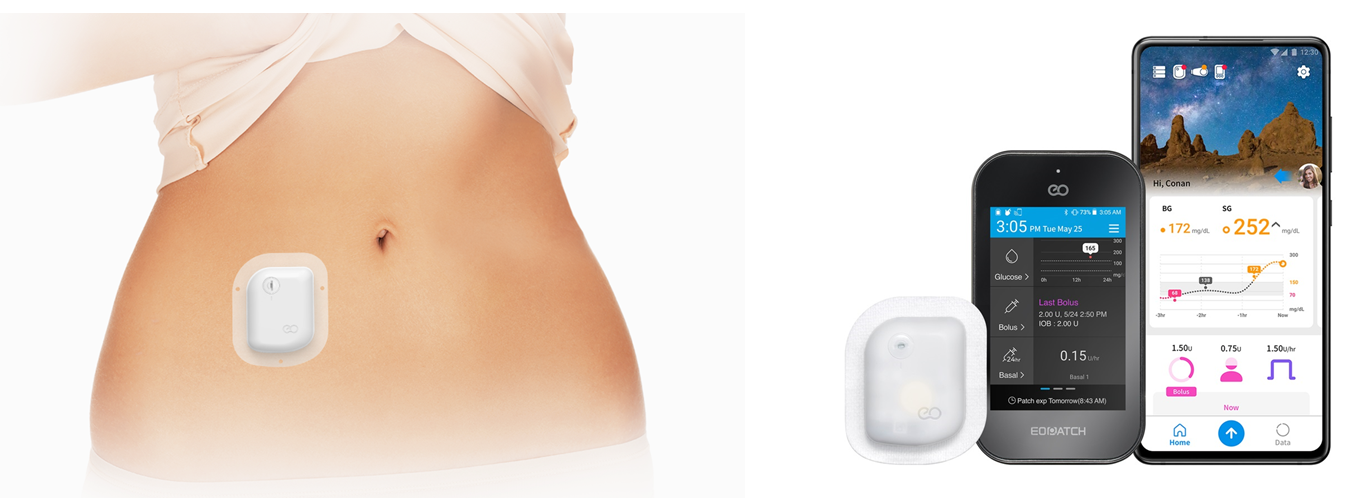
Unlike conventional insulin pumps, EOPatch can be attached to subcutaneous fat areas of the body such as the stomach area, arms, waist, and thighs, and can control the insulin delivery with a separate controller or your smartphone. In addition, as the device is tubeless, patients have more freedom of movement. Because EOPatch is not only small and lightweight, but also can be hidden underneath clothing, it is possible to keep your privacy as a diabetes patient from the world, which will definitely enhance the patient’s quality of life. Each EOPatch can provide up to 3.5 days (84 hours) of continuous insulin delivery, which allows for twice-a-week compliance, improving user compliance. The device can also withstand water for 24 hours.
「Narsha, the world’s first smartphone application for controlling wearable insulin pump」
We have recently launched the smartphone application “Narsha” for EOPatch users to control and monitor insulin infusion. In lieu ofa separate controller, users can simply access the smartphone application to control and monitor how much insulin EOPatch will deliver. Narsha is the first of its kind in the world. The application can communicate with certain blood glucose meters (BGM) via Bluetooth, providing the convenience of monitoring one’s blood glucose level and making insulin delivery decisions in one app. Also, EOBridge, a cloud-based diabetes data management program, is available in the app so patients can check the insulin delivery records on a smartphone and use the records as reference when they see their doctor. This will make it easier and more convenient for patients to manage diabetes. We have opened EOPatch online education center for users to learn how to use EOPatch. For more information, please visit: www.eopatch.com.
「Accelerating the commercialization of a two-part and an integrated wearable artificial pancreas systems」
In addition to EOPatch, we have recently completed the development of “EOPatch X,” a two-part wearable artificial pancreas (AP) system which integrated a wearable insulin pump “EOPatch” with a continuous glucose monitoring sensor (CGMS) and a closed loop pump control algorithm, and began a clinical study. We plan on launching the product in 2022. We are also making progress in developing “EOPancreas,” a fully integrated wearable artificial pancreas (AP) system that combines the “EOPatch”, a built-in glucose sensor, and an AP algorithm with an aim to launch it in 2023. Our artificial pancreas devices can monitor the blood glucose level and automatically adjust the insulin delivery to maintain optimal control of blood glucose levels for diabetic subjects.
Do you have any plans to enter the foreign market? What are your future business directions or blueprint?
「Focusing on the main market while seeking opportunities in emerging markets」
We have introduced our products to Europe for the first time in 2021. In addition to the major countries in Europe, we are also preparing ourselves to enter other global markets including China and the Middle East. Based on our technology developed after years of research and development, and quality verified in the Korean market, we will rapidly expand to the global market with a goal to reach 10 billion won in sales by 2022.